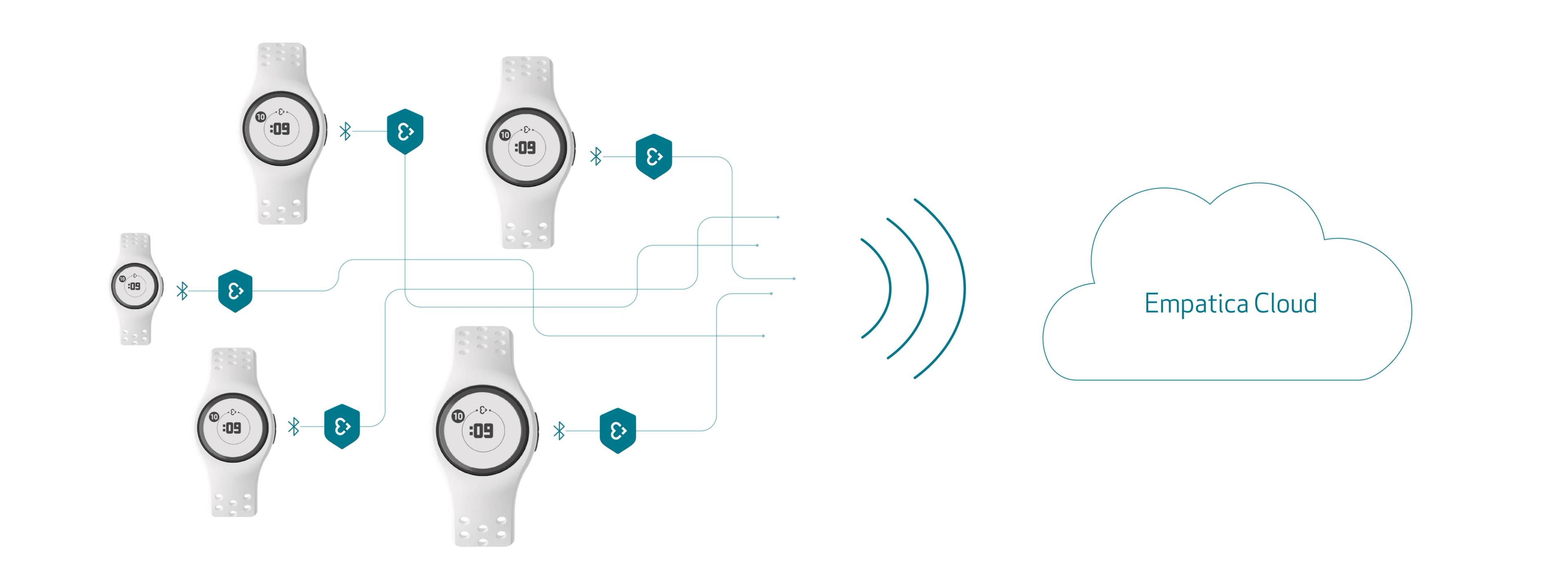
After only a month from winning the CE mark for both Aria and Empatica Care, we’re pleased to announce that Empatica’s EmbracePlus wearable has received the European CE mark as a Class lla medical device, for its ability to consistently provide quality physiological parameters to its users.

Designed and engineered to be the world’s most powerful medical smartwatch, the EmbracePlus combines the best of Empatica’s existing wearables: our FDA-cleared Embrace2, and the used in It continuously and remotely collects and processes key physiological signals from the wrist, including pulse rate, pulse rate variability, blood oxygenation, respiratory rate, skin temperature, electrodermal activity, rest, and actigraphy data. The data are continuously sent to a smartphone app and from there, they are transferred to the cloud for review and analysis by clinical researchers and healthcare professionals through a single dashboard. Data can be streamed simultaneously from thousands of devices. This is possible via or via third party-applications that can be developed.