Enrich digital biomarker data with eCOA
Now directly available with the Empatica Health Monitoring Platform
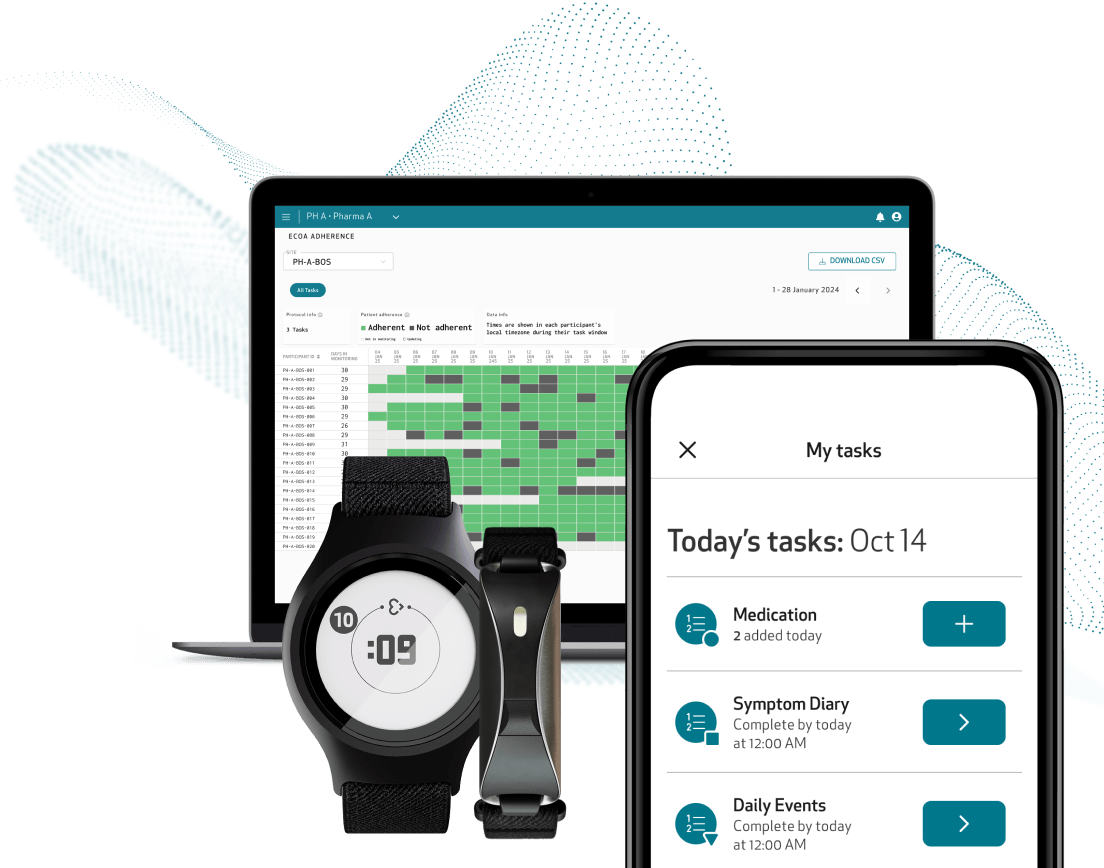
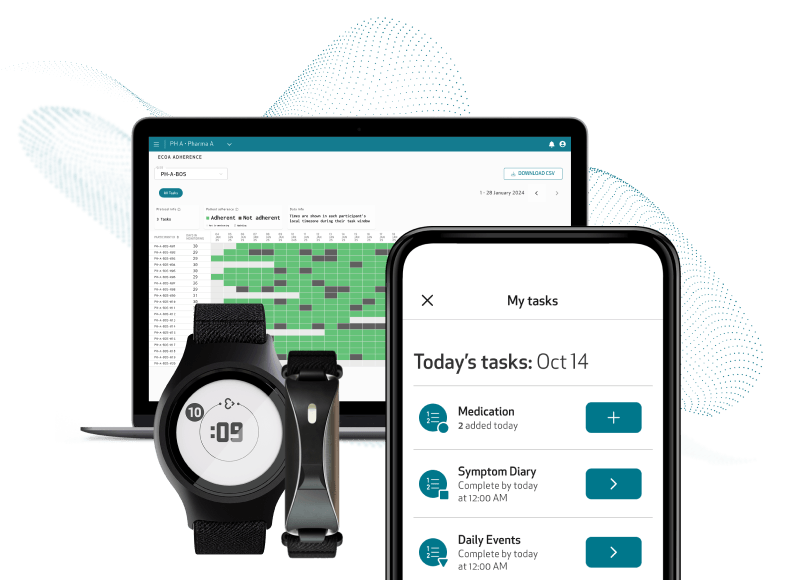


Low patient burden
Patients interact with only one app and device
Intuitive design
Familiar UX reduces patient and site training and improves adherence
Guided workflows
Clear step-by-step instructions for each test or questionnaire
Real-time feedback
Automatic progress tracking and compliance reminders
A richer set of data from participants
Global readiness
Regulatory-grade
End-to-end compliance
Unified data capture from a single platform
Ready to deploy
Zero friction for patients or sites


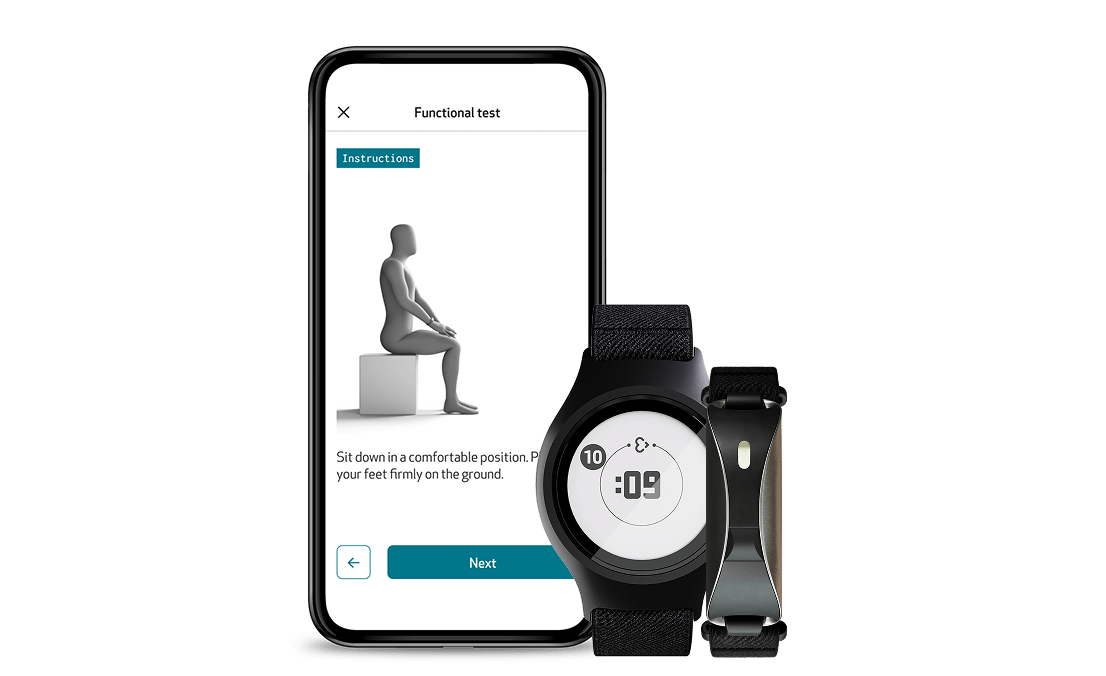
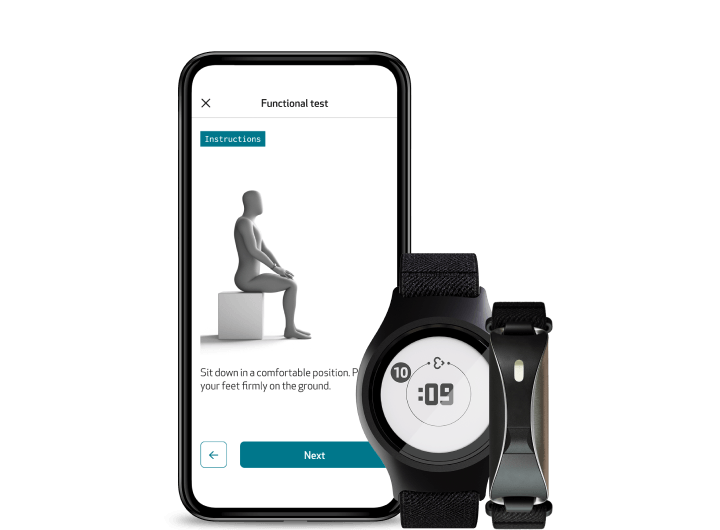
Functional Tests: Perform objective, repeatable assessments with EmbracePlus and EmbraceMini
Through the Care App, participants can perform study-specific functional tests remotely via guided flows with clear instructions, visuals, and reminders. Combined with continuous data from Empatica wearables, these tests deliver a richer, more objective view of each patient’s functional status. Functional Tests close the gap between episodic clinical evaluations and continuous, objective measurement, allowing trials to monitor patient performance from home with unprecedented fidelity.
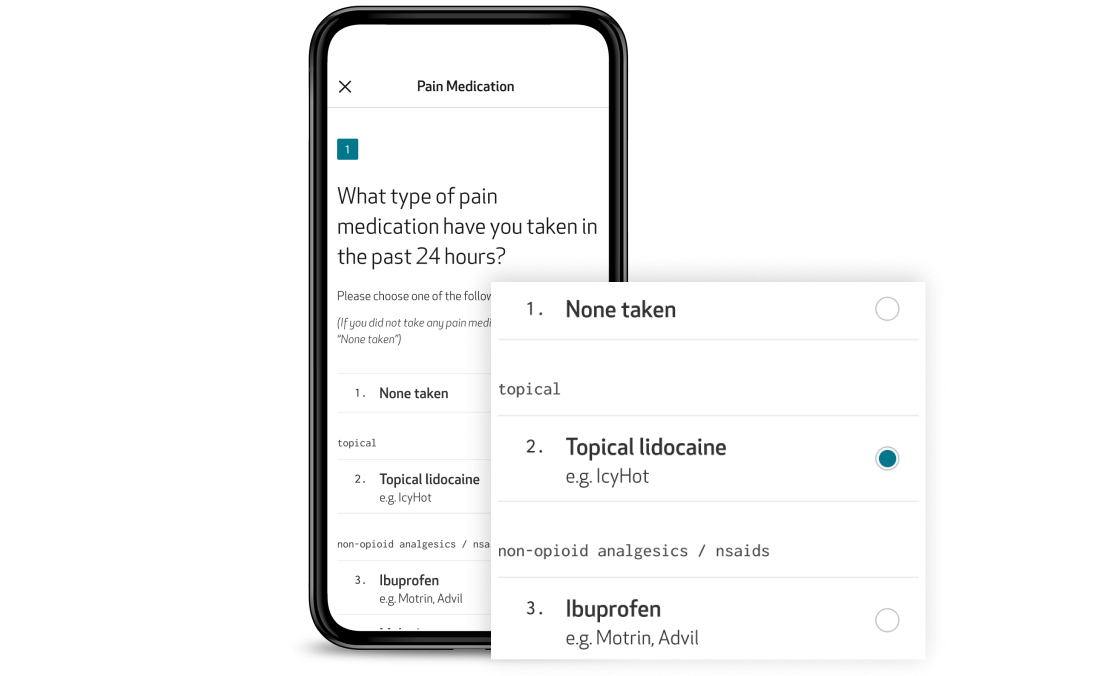
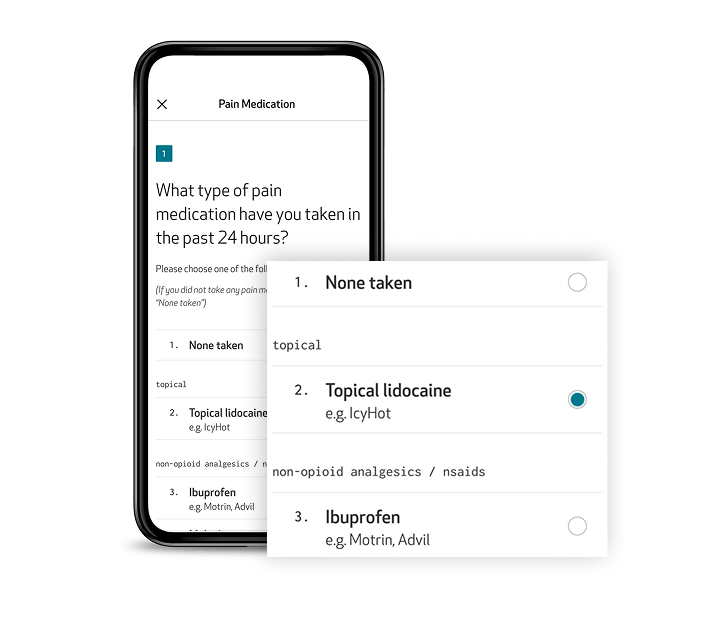
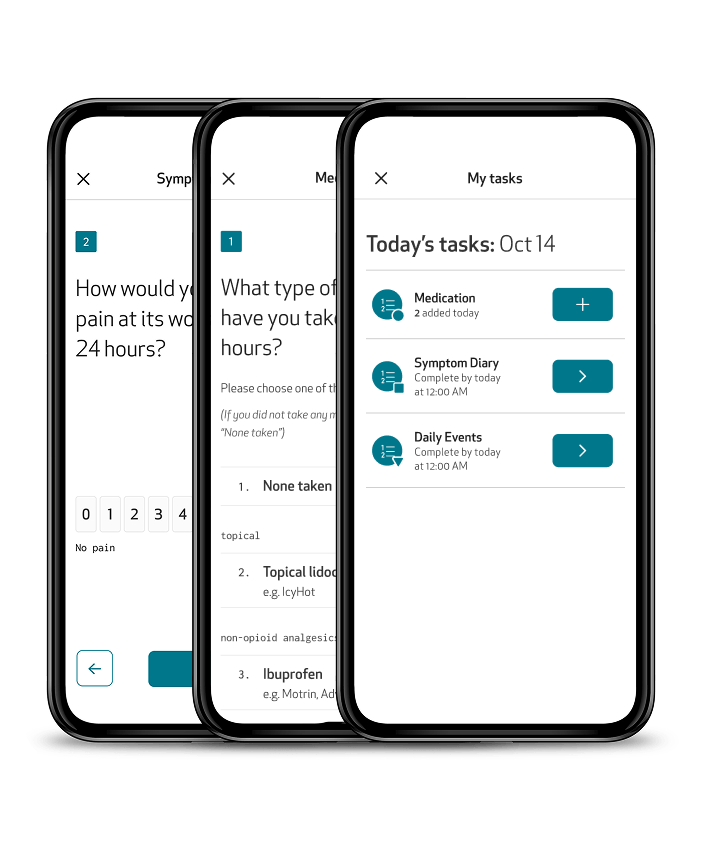
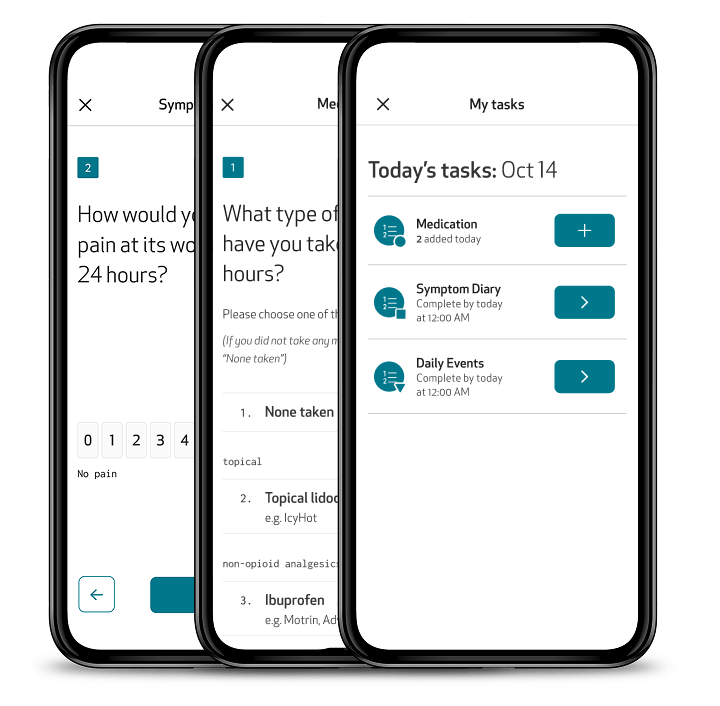
ePRO: Patient insights, captured directly and digitally
Capture subjective symptom and event data, such as pain, medication use, and daily events, directly within the Care App. Designed for accessibility and ease of use, the app guides patients through their ePROs with clear instructions, reminders, and intuitive navigation. Questionnaires can be completed directly from the app in the patient’s preferred language, even offline, ensuring continuous data collection with minimal burden.
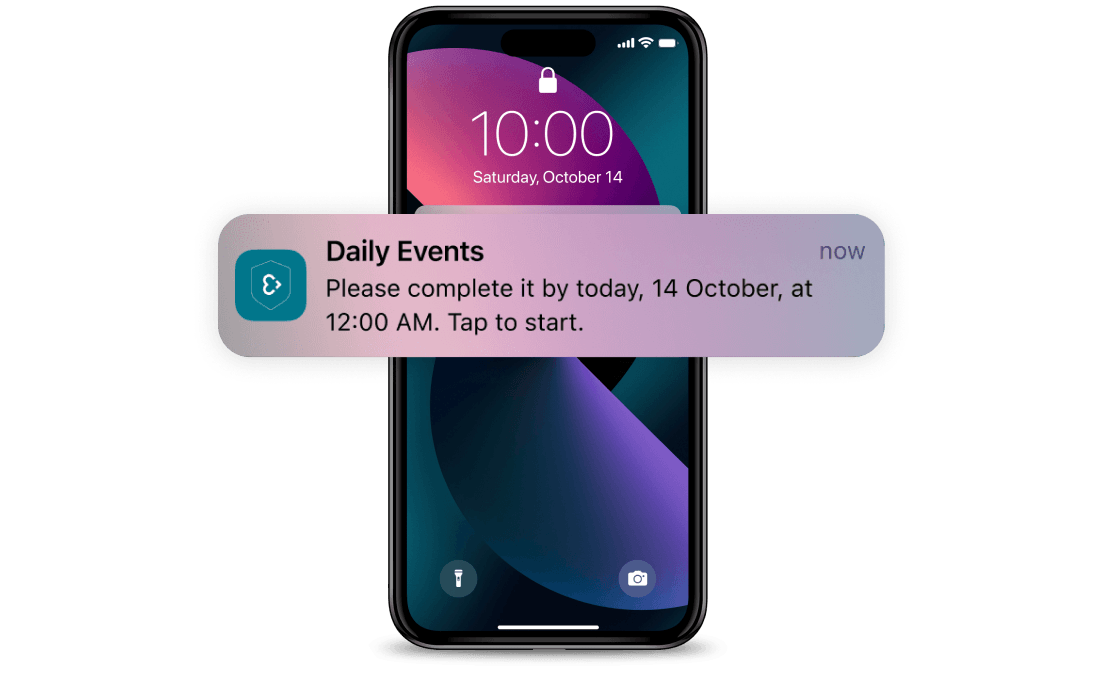

Medication reminders
Enable automated medication reminders via the Care app, addressing both the need of patients to be reminded to take their medication, and the need of clinicians to verify their adherence to treatment plans and trial protocols.


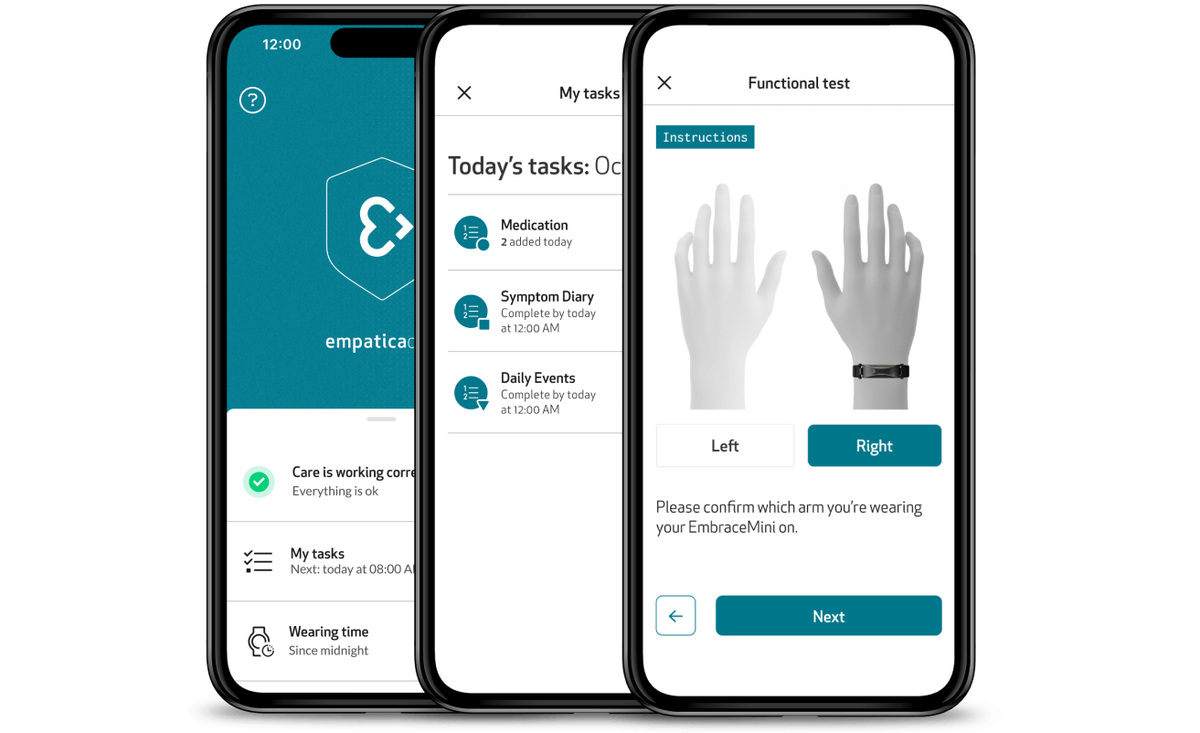
Designed for compliance, comfort, and clarity
All eCOA modules are directly integrated into the Care App, providing a single tool for patients for trial participation, data sharing, and system troubleshooting.
- Low burden: Patients interact with only one app and device
- Intuitive design: Familiar UX reduces patient and site training and improves adherence
- Guided workflows: Clear step-by-step instructions for each test or questionnaire
- Real-time feedback: Automatic progress tracking and compliance reminders


