Digital endpoints & wearable monitoring for Parkinson’s and Movement Disorder Trials





Digital endpoints for Parkinson’s clinical trials
Parkinson’s disease is defined by its cardinal motor symptoms: bradykinesia, tremor, rigidity, and postural instability, which are key biomarkers in clinical trials. Digital endpoints are increasingly used across trials on dopaminergic therapies (e.g. levodopa), disease-modifying treatments, and deep brain stimulation (DBS) and neuromodulation.
Key digital measures in Parkinson’s monitoring include:
- Bradykinesia
- Dyskinesia
- Sleep disturbances
- Wake After Sleep Onset (WASO)
- Tremor
- OFF time prediction
- Gait & Postural Stability
- Freezing of Gait
- Motor fluctuations
- Time spent immobile


Missing real-world context
Intermittent data collection
Clinician and patient subjectivity
Patient burden


Effectiveness of wearable devices in monitoring Parkinson’s Disease symptoms
Wearable devices equipped with accelerometers and gyroscopes used in Parkinson’s research have demonstrated strong potential in accurately and continuously monitoring key symptoms such as tremor, bradykinesia and dyskinesia.
- 95% sensitivity and 88% specificity comparing BKS with UPDRS III (Unified Parkinson's Disease Rating Scale).¹
- Highly significant correlation (R = 0.80, p < 0.0001) between DKS and AIMS (Abnormal Involuntary Movement Scale).¹
92.5% sensitivity and 92.9% specificity identifying tremor (PTT ≥ 0.8%).² - Wearable devices identify uncontrolled symptoms 72% of the time.³
Accelerometry data from wrist-worn sensors can measure step count in PD with <3% error rate.⁴
See publications
1. Griffiths RI, Kotschet K, Arfon S, et al. Automated assessment of bradykinesia and dyskinesia in Parkinson’s disease. J Parkinsons Dis. 2012;2(1):47-55.
2. Braybrook M, O’Connor S, Churchward P, et al. An Ambulatory Tremor Score for Parkinson’s Disease. J Parkinsons Dis. 2016;6(4):723-731.
3. Woodrow H, Horne M, Fernando C, Kotschet K, Treat to Target Study Group. A blinded, controlled trial of objective measurement in Parkinson’s disease. NPJ Parkinson’s Disease. 2020;6:35. https://doi.org/10.1038/s41531-020-00136-9
4. Shokouhi N, Khodakarami H, Fernando C, et al. Accuracy of Step Count Estimations in Parkinson’s Disease Can Be Predicted Using Ambulatory Monitoring. Front Aging Neurosci. 2022;14:904895. https://doi.org/10.3389/fnagi.2022.904895
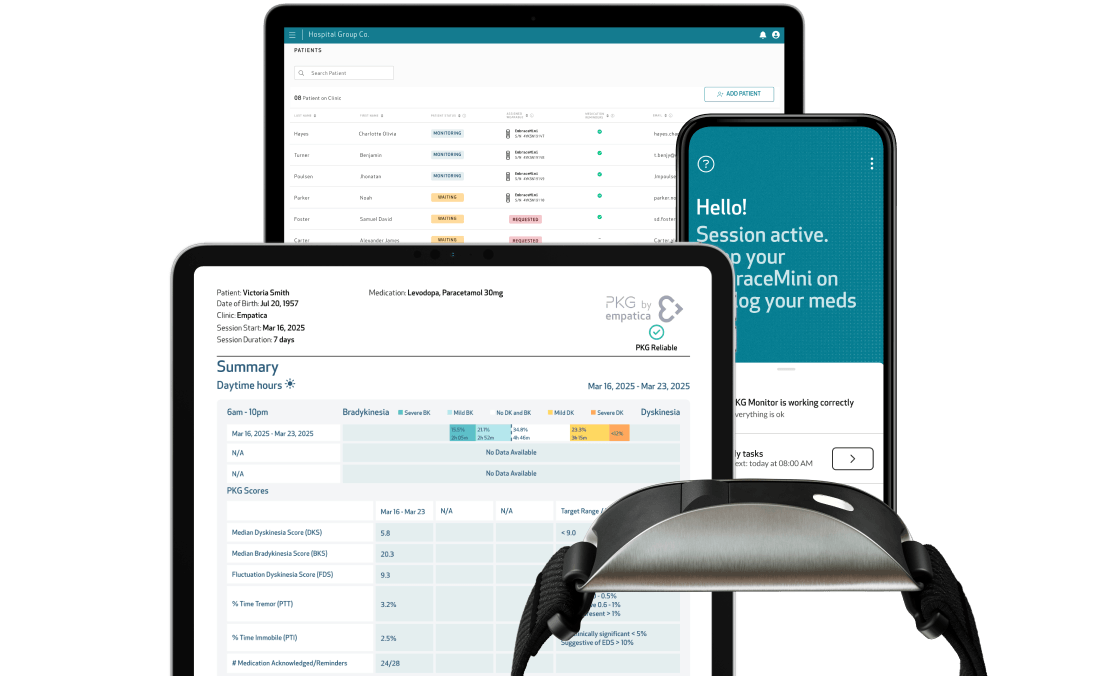

Enabling scalable remote monitoring for Parkinson’s trials
Empatica provides a clinical-grade wearable platform designed to support continuous, remote monitoring in neurological clinical trials.
- FDA-cleared and CE-marked platform, devices and apps
- Continuous actigraphy and movement analysis for motor symptom tracking
- Continuous data collection for up to 14 days
- High patient adherence through comfortable, wrist-worn design
- 38 validated movement disorder measures
- Over 200 sleep and activity measures
- Secure data infrastructure supporting compliance requirements (GDPR, HIPAA)
Learn about our Clinical Trials offering
What are relevant digital endpoints in Parkinson’s clinical trials?
Are wearable devices validated for Parkinson’s research?
How are movement disorders traditionally measured in Parkinson’s trials?
What are the benefits of continuous monitoring in Parkinson’s disease?
Can digital biomarkers replace clinical scales?
How effective are wearable devices in monitoring Parkinson's symptoms?




